Science
She Lobbied for Formaldehyde. Now She’s at E.P.A. Approving New Chemicals.

Formaldehyde, the chemical of choice for undertakers and embalmers, is also used in products like furniture and clothes. But it can also cause cancer and severe respiratory problems. So, in 2021, the Environmental Protection Agency began a new effort to regulate it.
The chemicals industry fought back with an intensity that astonished even seasoned agency officials. Its campaign was led by Lynn Dekleva, then a lobbyist at the American Chemistry Council, an industry group that spends millions of dollars on government lobbying.
Dr. Dekleva is now at the E.P.A. in a crucial job: She runs an office that has the authority to approve new chemicals for use. Earlier she spent 32 years at Dupont, the chemical maker, before joining the E.P.A. in the first Trump administration.
Her most recent employer, the chemicals lobbying group, has made reversing the Environmental Protection Agency’s course on formaldehyde a priority and is pushing to abolish a program under which the agency assess the risks of chemicals to human health. In recent weeks it has urged the agency to discard its work on formaldehyde entirely and start from scratch in assessing the risks.
The American Chemistry Council is also seeking to change the agency’s approval process for new chemicals and speed up E.P.A.’s safety reviews. That review process is a key part of Dr. Dekelva’s purview at the agency.
Another former chemistry council lobbyist, Nancy Beck, is back alongside Dr. Dekleva at the E.P.A. in a role regulating existing chemicals. The council’s president, Chris Jahn, told a Senate hearing shortly after the Trump inauguration that his group intended to tackle the “unnecessary regulation” of chemicals in the United States. “A healthy nation, a secure nation, an economically vibrant nation relies on chemistry,” he said.
It is not unusual or unlawful for industry groups to seek to influence public policy in the interest of their member companies. The A.C.C. estimates that products using formaldehyde support more than 1.5 million jobs in the United States.
What has been extraordinary, health and legal experts said, is the extent of the industry’s effort to block the E.P.A.’s scientific work on a chemical long acknowledged as a carcinogen, and how the architect of the effort was back at the agency as a regulator of chemicals. At the same time, the Trump administration has moved to sharply reduce the federal scientific work force.
“They already have a track record of ignoring the science,” said Tracey Woodruff, director of the Program on Reproductive Health and the Environment at the University of California, San Francisco. “Now, they’re in charge of government agencies that decide the rules.”
While leading the chemistry council’s fight to limit formaldehyde regulation, Dr. Dekleva called for investigations of federal officials for potential bias. The industry group used freedom of information laws to obtain emails of federal employees and criticized them in public statements for what they had written. It submitted dozens of industry-funded research papers to agencies that minimized the risks of formaldehyde.
The A.C.C. also sued both the E.P.A. and the National Academies, which advises the nation on scientific questions, accusing researchers of a lack of scientific integrity.
Allison Edwards, a chemistry council spokeswoman, said officials from the group had regularly met with E.P.A. staff members “to share critical science and to try and ensure an assessment of any chemistry is objective, employs rigorous scientific standards, and is reflective of real-world human exposure.” She said, “We’re asking to be one of many stakeholders at the table.”
Molly Vaseliou, a spokeswoman for the E.P.A., said the agency would continue to make sure it “ensures chemicals do not pose an unreasonable risk to human health or the environment.” At the same time, the agency would also work to approve “chemicals that are needed to power American innovation and competitiveness,” she said.
Formaldehyde’s cancer risk
Formaldehyde’s fumes can cause wheezing and a burning sensation in the eyes, especially when they accumulate indoors. That danger was apparent when formaldehyde in plywood used to build temporary trailer homes for victims of Hurricane Katrina sickened dozens of people.
And there are longer-term dangers, namely several types of cancers. The World Health Organization’s International Agency for Research on Cancer concluded in 2004 that the chemical is a human carcinogen, and the U.S. Department of Health listed it as a human carcinogen in 2011.
The chemical is restricted in the workplace, in certain composite wood products, and in pesticides. Yet efforts to strengthen overall regulations in the United States have stalled in the face of industry opposition.
President Biden, whose “cancer moonshot” program had made reducing cancer deaths a priority, revived in 2021 an E.P.A. assessment of the health effects of the chemical, and published a draft the following year. That effort, under the agency’s Integrated Risk Information System, was the first step toward regulating formaldehyde.
The chemistry council led a coalition of industry groups, including the Composite Panel Association and Kitchen Cabinet Manufacturers, arguing that formaldehyde had already been rigorously studied and that strict industry controls were in place.
In a half-dozen letters to the E.P.A., Dr. Dekleva, on behalf of a formaldehyde panel at the industry group, raised a list of complaints about the way the agency was carrying out its assessment.
She questioned research linking formaldehyde to leukemia, or cancer of the blood, and accused the agency of not relying on the best available science. There was a dose, she said, at which formaldehyde did not cause risk. There was also research, she said, that showed inhaled formaldehyde did not easily travel beyond the nose to cause harm to the body.
In light of these issues, Dr. Dekleva wrote, agency’s draft assessment was “flawed and unreliable without significant revision.”
To bolster its case, the industry group enlisted experts at consulting firms to submit opinions and studies to the E.P.A. minimizing formaldehyde’s risks. The firms included those previously commissioned by tobacco companies to help defend cigarettes.
The A.C.C. also submitted 41 peer-reviewed studies that it said refuted a link between formaldehyde and leukemia. A New York Times review found that the majority of the studies were funded by industry groups, including at least 11 from the Research Foundation for Health and Environmental Effects, an organization established by the American Chemistry Council.
David Michaels, an epidemiologist and professor at George Washington University School of Public Health and assistant secretary of labor under President Barack Obama, said the industry strategy was to create the appearance of disagreement among scientists.
While it’s true, he said, that inconsistencies can always exist in studies on humans, “there’s little disagreement among independent scientists that formaldehyde causes cancer.”
Scientists targeted
For more than 150 years, the National Academies has advised the U.S. government on science. In 2021, it was asked to weigh in on the E.P.A.’s work on formaldehyde.
It became a target of the American Chemistry Council.
The industry group used freedom of information laws to obtain internal emails of members and support staff of a panel assessing the E.P.A.’s formaldehyde review, and it accused one staff of showing “bias in favor of disputed research claiming formaldehyde causes leukemia.”
The staff member, a former Environmental Protection Agency scientist, had for example described as “wonderful” the news that Congress might try to replicate an influential Chinese study that had shown formaldehyde could cause leukemia.
Wendy E. Wagner, professor at the University of Texas School of Law and an expert on the use of science by environmental policymakers, said she did not see how the comment reflected bias. “After all, they don’t know what the results will be, do they?” she said. “I would expect all scientists to be enthusiastic about potential future research.”
Dr. Dekleva called for investigations at both the E.P.A. and the National Academies, and for the removal of potentially biased panel members and staff. That included scientists who had previously accepted federal research grants.
In July 2023, the industry group sued the E.P.A., as well as the National Academies, accusing researchers of a lack of scientific integrity. The chemistry council said that lack of integrity made the use of the National Academies research in regulating formaldehyde “arbitrary, capricious, and unlawful.”
“It was relentless, and beyond the pale,” said Maria Doa, a scientist at the E.P.A. for 30 years who is now senior director of chemicals policy at the Environmental Defense Fund. “They really ratcheted up their attacks on federal employees.”
The National Academies stood its ground, issuing a report the following month affirming the E.P.A.’s Integrated Risk Information System findings that formaldehyde is carcinogenic and increases leukemia risk.
Those conclusions are shared by other global health authorities.
Mary Schubauer-Berigan, the evidence-synthesis head at the World Health Organization’s Agency for Research on Cancer, said there was “sufficient evidence in humans” that formaldehyde causes leukemia as and nasopharynx cancer. Mikko Vaananen, a spokesman for the European Chemicals Agency, said that while some questions around specific links to leukemia remained unanswered, evidence was sufficient to classify formaldehyde as a carcinogen. Formaldehyde “cannot in principle be placed on the E.U. market,” he said.
In March 2024, a federal judge dismissed the chemistry council’s lawsuit. And early this year, near the end of the Biden administration, the E.P.A. issued a final risk determination, under the Toxic Substances Control Act: Formaldehyde “presents an unreasonable risk of injury to human health.”
Mary A. Fox, an expert in chemical risk assessment at the Johns Hopkins University Bloomberg School of Public Health and a member of a committee that reviewed the E.P.A.’s research on formaldehyde, said agency scientists had accurately reflected the uncertainties around the links between formaldehyde and leukemia. But they had documented many other streams of evidence that indicated that link, Dr. Fox said.
“It’s an inevitable progress of science, that as we learn more over time, we generally learn that health effects appear at lower concentrations than we had thought,” she said.
Following Mr. Trump’s re-election, the American Chemistry Council signed onto a letter from a range of industry groups calling for broad changes to policy, specifically citing formaldehyde. “We urge your administration to pause and reconsider” the E.P.A. findings on formaldehyde, the Dec. 5 letter said.
The E.P.A. “should go back to the scientific drawing board,” chemistry council said in January. The group was particularly concerned about the workplace limits the agency was suggesting, which it said ignored steps companies were already taking to protect workers, like the use of personal protective equipment.
The A.C.C. is also supporting a bill from Republican members of Congress that would end the Integrated Risk Information System.
Soon after, Trump transition officials said Dr. Dekleva would be returning to the E.P.A. to run a program assessing chemicals for approval. The chemistry council, which has long complained of a backlog, is pushing the agency to speed up approvals.
During the first Trump administration, agency whistle-blowers described in an inspector general’s investigation how they had faced “intense” pressure to eliminate the backlog, sometimes at the expense of safety. Shortly after the inauguration, the Trump administration fired the inspector-general who carried out the investigation.
On Jan. 20, the A.C.C. welcomed President Trump. “Americans want a stronger, more affordable country,” said Mr. Jahn, the group’s president. “America’s chemical manufacturers can help.”

Science
Oxnard man smuggled baby crocodiles, among 1,700 reptiles, gets 5 years

An Oxnard man has been sentenced to more than five years in prison for smuggling at least 1,700 reptiles worth more than $739,000 into the U.S. over six years, the U.S. Department of Justice announced Friday.
The animals, including baby crocodiles and Yucatán box turtles, were bought and sold over social media and came from Mexico, Hong Kong and elsewhere, an investigation led by the U.S. Fish and Wildlife Service revealed.
From January 2016 to February 2022, Perez and co-conspirators brought in wild animals without the permits required by the Convention on International Trade in Endangered Species of Wild Fauna and Flora — and without declaring them, the Justice Department said.
In August 2022, Jose Manuel Perez pleaded guilty in federal court to one count of smuggling goods into the country and one count of wildlife trafficking.
The animals smuggled from Mexico were advertised on social media, with defendants posting photos and videos of the reptiles being captured in the wild.
People working with Perez would collect the reptiles including Mexican box turtles and Mexican beaded lizards, at from an airport in Ciudad Juárez, then move them by car over the border to El Paso.
According to federal authorities, Perez paid people a “crossing fee” each time they traversed the border. Payment depended on how many animals they trafficked, the size of the package and the level of risk they faced.
Sometimes Perez and another person would traveled to Mexico to buy animals taken from the wild to smuggle into the U.S. Once shipped, they were transported to Perez’s home, in Missouri and then California after he moved there.
When the sentence came down, Perez was already serving nine years for felony possession of firearms. Due to convictions in Ventura County Superior Court for “street terrorism” and assault with a deadly weapon, he is not allowed to have firearms, the department said.
According to the San Diego Zoo Wildlife Alliance, illegal wildlife trafficking is the second-largest threat to species after habitat loss and the world’s fourth-most-lucrative trafficking industry.
“Illegal wildlife trafficking not only diminishes the populations of targeted wildlife species, it also impacts related species, their interconnected ecosystem, local and global economies, and has the potential to impact the health of people through zoonotic disease transmission,” the alliance says on its website.
Reptiles get caught in the fray. Earlier this month, the Justice Department announced that a Daly City man suspected of purchasing and exporting hundreds of poached turtles from Florida was facing federal wildlife trafficking charges.
The U.S. attorney’s office for the Southern District of California and a section of the Justice Department’s Environment and Natural Resources Division, along with U.S. Customs and Border Protection and Homeland Security Investigations, assisted federal wildlife officials with the investigation into Perez’s dealings. The case was prosecuted in U.S. District Court for the Central District of California.
Science
Video: Blue Origin Rocket Explodes on Florida Launchpad

new video loaded: Blue Origin Rocket Explodes on Florida Launchpad
transcript
transcript
Blue Origin Rocket Explodes on Florida Launchpad
A rocket built by the Jeff Bezos-owned space company, Blue Origin, blew up during a test at the Cape Canaveral Space Force Station in Florida.
-
“Oh, no, that’s an explosion.” (explosion erupts) “That is crazy.” “What?” “Oh, my God!”

By Nailah Morgan
May 29, 2026
Science
Parental mental health — not medication — drives autism correlation, new study finds
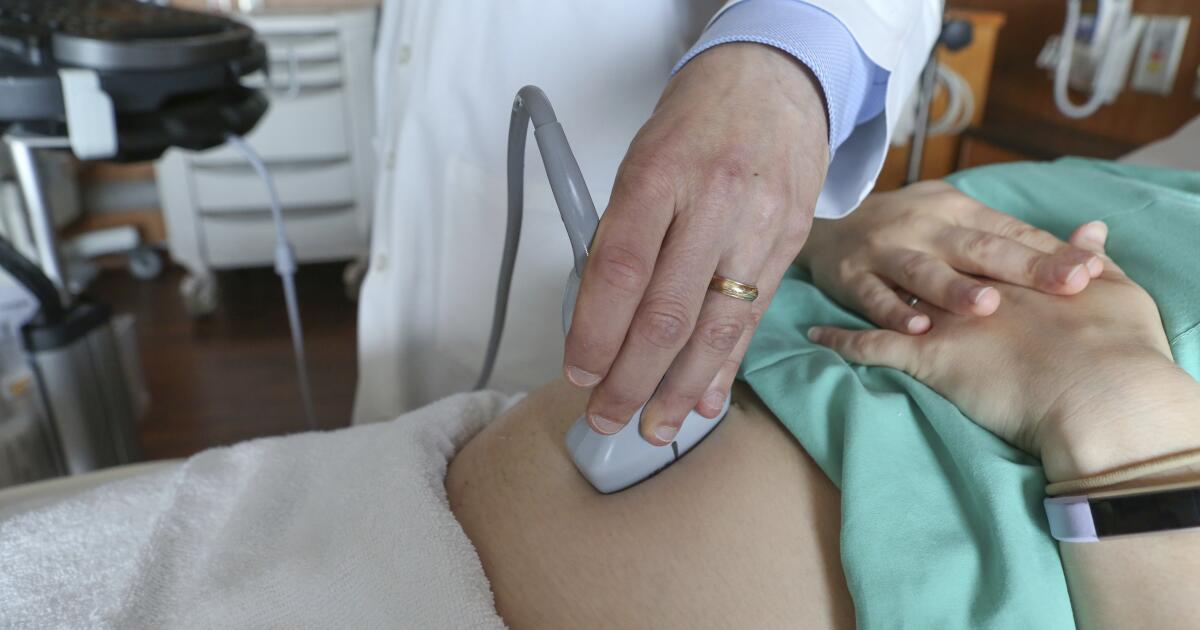
A sweeping new review of prenatal antidepressant use underscores a finding that has surfaced repeatedly throughout the last decade: While parental depression is strongly linked to child neurodevelopmental disorders, taking antidepressants during pregnancy does not appear to significantly increase a child’s risk of autism.
In an analysis of 37 separate studies covering more than 25 million pregnancies, a research team from the University of Hong Kong found that children born to women who took antidepressants while pregnant were indeed more likely to later be diagnosed with autism or attention-deficit/hyperactivity disorder (ADHD).
But when the researchers took into account confounding factors such as a family history of neurodevelopmental disorders or mothers’ preexisting mental health conditions, the correlation disappeared.
The data showed that children born to women with a history of depression were more likely to be diagnosed with autism or ADHD, regardless of whether their mother took psychiatric medication. Children were also more likely to be diagnosed with autism and ADHD if their fathers took antidepressants during their gestation, even if their mothers did not — an association that suggests a genetic link, not a pharmacological one.
The results were published this month in the journal the Lancet.
“Our findings are consistent with current clinical guidelines, which generally support continuing antidepressant treatment during pregnancy when it is clinically indicated,” said Dr. Wing-Chung Chang, a psychiatry professor at the University of Hong Kong and the paper’s senior author. “Our findings do not provide strong evidence that prenatal antidepressant exposure causes neurodevelopmental disorders.”
The possibility that antidepressant use in pregnancy may play a role in neurodevelopmental conditions has been a source of anxiety for many expectant parents since at least 2015, when a much-publicized Canadian study observed that women who took certain antidepressants later in pregnancy were about twice as likely to have an autistic child than women who did not take the drugs.
Multiple studies since then have also identified a correlation between a woman’s use of antidepressants during pregnancy and her child’s later diagnosis of autism, and to a lesser extent, ADHD.
But ending the analysis there overlooks a crucial distinction, researchers say: the possibility that the association actually is between the neurodevelopmental disorders and depression, not the medication.
Autistic people of all ages are significantly more likely than their neurotypical peers to be diagnosed with mental health conditions such as depression or anxiety. Large-scale population studies have found that autistic adults are up to three times as likely to have depression compared with non-autistic people.
The reasons for mental health symptoms in autistic people are varied and complex, and the challenges of navigating a world designed for a different way of thinking may play an important role. But research has also identified multiple genetic profiles and biological pathways common to autism and mood disorders, and it’s likely that both conditions are at least partially the result of family genetics.
“The mental health of your family tree is in some way statistically associated with your risk of autism,” said Brian K. Lee, a professor of epidemiology and biostatistics at Drexel University.
Neither depression nor autism causes the other. Lee compared their frequent co-occurrence to the pairing of fiery red hair and pale, sunburn-prone skin: two highly heritable traits that can easily occur independently in a given individual, but that often travel together through family trees.
“What the literature has shown us so far is that while there does, at face value, appear to be an association of slightly increased risk of autism in mothers who take antidepressant medications, when you control for the underlying depressive disorder that risk goes away,” said Dr. Kathryn Erickson-Ridout, a senior psychiatrist for the Permanente Medical Group and research scientist with the Kaiser Permanente Division of Research. “This evidence shows us that most likely, the biological pathways that are disrupted in major depression are also important for autism.”
Erickson-Ridout compared the chilling effect of the 2015 Canadian study on psychiatric care for pregnant women with the anxiety around vaccines sparked by Andrew Wakefield’s since-retracted 1998 paper inaccurately linking autism to the mumps, measles and rubella shot.
The Canadian study did not contain major errors as Wakefield’s paper did, though some critics argued at the time that it didn’t sufficiently control for confounding factors such as maternal depression.
But its media coverage often failed to make clear both the low overall risk of autism — 1.2% of babies born to women who took selective serotonin reuptake inhibitors during their second or third trimester were later diagnosed with autism, compared with 0.7% of babies in the general population — or weigh the risk of antidepressant use against the risks of untreated depression.
Its effects persist today. Last year, the U.S. Food and Drug Administration convened a controversial panel on prenatal SSRI use. Nine of the panel’s 10 members were researchers, doctors or psychologists who have previously questioned the drugs’ safety or criticized antidepressant use in general. Among them was Anick Berard, an epidemiologist and lead author of the 2015 Canadian paper.
Suicide is the second-leading cause of maternal mortality in the U.S., with homicide being the first.
Any discussion of the risks of antidepressant medications has to be weighed against the potential harms of abruptly ceasing or refusing to treat a potentially life-threatening mental health condition, said Dr. Katie Unverferth, a reproductive psychiatrist and medical director of UCLA’s Maternal Mental Health Program.
“Pregnancy is such an anxious time at baseline — so many new things are happening, and your body’s changing, and you want to make sure you’re doing the right thing for yourself and your developing baby,” Unverferth said. “This study just provides additional reassuring data.”
-

 Science43 seconds ago
Science43 seconds agoOxnard man smuggled baby crocodiles, among 1,700 reptiles, gets 5 years
-

 Sports7 minutes ago
Sports7 minutes agoA new board game mocks Shai Gilgeous-Alexander for ‘foul baiting.’ He wants it destroyed
-

 World19 minutes ago
World19 minutes agoIsrael, Russia among new additions on UN sexual violence ‘blacklist’
-

 News49 minutes ago
News49 minutes agoWhich first lady feared her husband might be having a stroke? The quiz knows
-

 Los Angeles, Ca2 hours ago
Los Angeles, Ca2 hours agoRescued sea lion pups released in Manhattan Beach
-

 Detroit, MI3 hours ago
Detroit, MI3 hours agoSunda New Asian brings bold flavors to Detroit
-

 San Francisco, CA3 hours ago
San Francisco, CA3 hours agoDriver Arrested After Pedestrian Killed, Three Injured In Mission District Crash
-

 Dallas, TX3 hours ago
Dallas, TX3 hours agoMcAllen Welcomes Texas Hockey | Dallas Stars











